Nasisoft

NASISOFT – SAUDZĪGAI DEGUNA GĻOTĀDAS KOPŠANAI
NASISOFT ir sterils 0.9% NaChl šķīdums kombinācijā ar hialuronskābi, piemērots lietošanai intranazāli un inhalāciju veidā visu vecumu pacientiem pie elpošanas ceļu saslimšanām iepilinot deguna ejās vai lietojot inhalācijās izmantojot smidzinātāju (nebulaizeri)
- Deguna dobuma ikdienas kopšanai
- Elpošanas ceļu gļotādas mitrināšanai
- Saudzīgai deguna gļotādas attīrīšanai
- Plaši izmantojams, intranazāli vai inhalācijās, augšējo elpošanas ceļu saslimšanu laikā (akūts vai hronisks rinīts, nazofaringīts, rinosinusīts u.c.)
- Akūtu un hronisku alerģisku rinītu laikā
Nasisoft atvieglo elpošanu, samazinot deguna un deguna blakusdobumu gļotādas tūsku un kairinājumu, kā arī veicina ātrāku krēpu izdalīšanos, hialuronskābe novērš antigēnu (alergēnu un mikroorganismu) adhēziju (pielipšanu) pie gļotādas, lietojams profilaktiski arī alerģiskā rinīta gadījumā.
Lietošana: piemērots lietošanai visu vecumu pacientiem pie elpošanas ceļu saslimšanām lietojot intranazāli vai inhalācijās izmantojot smidzinātāju (nebulaizeri)
Sastāvs. 1ml šķīduma satur:
- Nātrija hialuronātu 0,1 mg;
- Nātrija hlorīdu 9,0 mg.
Iepakojums: 4ml x N10 polimēra konteineri ar sterilu šķīdumu
Kur pirkt?
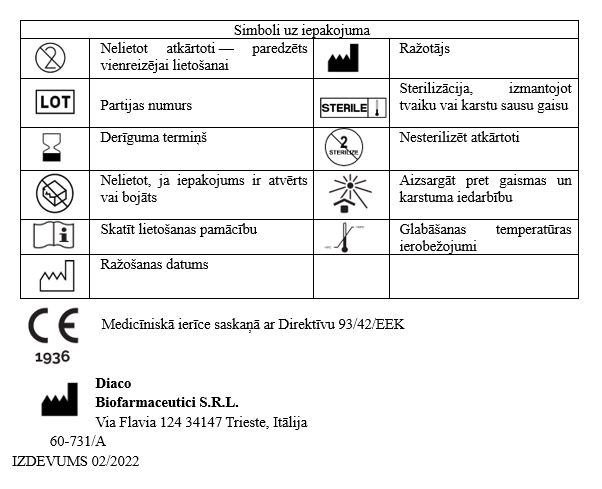
Medicīniskās ierīces NASISOFT lietošanas instrukcija
NASISOFT Sterils šķīdums inhalācijām un intranazālai ievadīšanai
Apraksts: NASISOFT ir sterils šķīdums inhalācijām ar smidzinātāju un/vai intranazālai ievadīšanai ar tiešu iepilināšanu no polimēra dozatora deguna ejās. Tas ir dabisks izotonisks sāls šķīdums tādā koncentrācijā, kas ir pielāgota saderībai ar šķidrumiem cilvēka ķermenī.
Lietošanas mērķis: NASISOFT šķīdumu lieto elpceļu mitrināšanai un kopšanai ikdienā, kā arī saaukstēšanās laikā ja ir tādi simptomi kā aizlikts deguns un klepus.
Sastāvs: Nātrija hlorīds — 9,0 mg/ml, Nātrija hialuronāts — 1,0 mg/ml, Sterils ūdens
Produkta klasifikācija
Sterilizēta un apirogēna IIb klases medicīniskā ierīce.
Primārais iepakojums: 4 ml iepriekš uzpildīts vienreizējas lietošanas polimēra dozators, katrs satur 0,9 % sāls šķīdumu un Nātrija hialuronātu 0.1%
Sekundārais iepakojums satur: 10 polimēra dozatorus 4ml ar sterilu NASISOFT šķīdumu; vai 60 polimēra dozatorus 4ml ar sterilu NASISOFT šķīdumu.
Tikai iepakojuma saturs ir sterils.
Medicīniskā ierīce ir sterilizēta ar tvaika sterilizāciju.
Indikācijas: NASISOFT šķīdums, saskaņā ar pamācību, piemērots lietošanai jaundzimušajiem, bērniem, grūtniecēm un sievietēm, kuras baro bērnu ar krūti, kā arī pieaugušajiem gadījumos, kuros ir indicēta šķīduma lietošana:
- deguna dobuma kopšanai ikdienā un lietošanai pacientiem ar deguna gļotādas slimībām, kuru pazīmes ietver deguna gļotādas sausumu vai gļotu veidošanos (atrofisks, alerģisks, infekciozs un zāļu izraisīts rinīts);
- deguna gļotādas mitrināšanai, kad telpās tiek izmantots gaisa kondicionieris vai apkures sezonas laikā, automašīnās ar gaisa kondicionētāju, kā arī citos gadījumos kad tiek veicināta elpceļu gļotādas sausināšana piem. lidojumu laikā;
- pēc deguna dobumā veiktām operācijām.
Lietošana: Inhalācijās. izmantojot inhalatoru/smidzinātāju, ievērojot ierīces ražotāja lietošanas norādījumus Ieteicamā deva ir viens 4 ml polimēra konteiners divas reizes dienā vai saskaņā ar ārsta rekomendācijām.
Intranazāla ievadīšana. Šķīduma intranazālu ievadīšanu var veikt, saspiežot atvērtu polimēra konteineru un ļaujot saturam ieplūst katrā deguna ejā. Lietot vienu 4 ml polimēra konteineru vienā lietošanas reizē, vienu līdz četras reizes dienā vai saskaņā ar ārsta rekomendācijām.
Lietošanas ilgums līdz 4 nedēļām vai saskaņā ar ārsta rekomendācijām.
Brīdinājumi un piesardzības pasākumi lietošanai:
- Šķīdums ir paredzēts lietošanai inhalācijās vai intranazālas ievadīšanas veidā. To nedrīkst injicēt.
- Nelietot, ja šķīdums nav caurspīdīgs un bezkrāsains.
- Nelietot pēc derīguma termiņa beigām.
- Nelietot, ja polimēra dozators ir atvērts vai bojāts.
- Atvērts polimēra dozators jāizlieto tūlītēji pēc atvēršanas.
- Katrs polimēra dozators ir paredzēts vienas reizes lietošanai. atlikušais neizlietotais daudzums vairs nav sterils.
- Nesterilizēt atkārtoti. Atkārtota sterilizācija var izraisīt savstarpēju piesārņošanos.
- Šķīdumu nedrīkst sasaldēt.
- Šķīdums nav paredzēts lietošanai iekšķīgi.
- Bērniem šķīdumu jālieto pieaugušo uzraudzībā.
- Medicīniskā ierīce satur sīkas detaļas, kas var radīt aizrīšanās risku. Uzglabājiet bērniem neredzamā un nepieejamā vietā.
Kontrindikācijas: NASISOFT šķīdums ir kontrindicēts gadījumos, kad ir individuāls paaugstināts jutīgums (hipersensitivitāte) pret jebkuru no produkta sastāvdaļām. Personām ar individuālu nepanesību pret atsevišķām šķīduma sastāvdaļām ir iespējamas alerģiskas reakcijas.
Izotoniskais sāls šķīdums var izraisīt atgriezenisku bronhu sašaurināšanos, ja to ieelpo ļoti jutīgi pacienti ar bronhiālo astmu vai hiperreaktīvu bronhu sistēmu. Īpaši jutīgām personām ārstēšanas sākumā ir ieteicama premedikācija ar bronhodilatatoriem, kas var palīdzēt novērst bronhu spazmas izraisošas reakcijas. Premedikācija ar bronhodilatatoriem jāveic ārsta uzraudzībā. Pārtrauciet preparāta lietošanu, ja rodas bronhu spazmas vai ilgstošs klepus, un informējiet par to ārstējošo ārstu.
Lietošanas norādījumi:
Inhalācijās, izmantojot smidzinātāju (nebulaizeri)
- Pirms lietošanas pārbaudiet uz primārā iepakojuma norādīto derīguma termiņu. Nelietot ja derīguma termiņš ir beidzies.
- Paņemiet vienu polimēra dozatoru, kas satur NASISOFT šķīdumu, no 10 polimēra dozatoru sloksnes.
- Atveriet polimēra dozatoru, pagriežot tā aizdari ar skrūvējošu kustību.
- Lietojiet šķīdumu izmantojot smidzinātāju, ievērojot ierīces ražotāja sniegto lietošanas pamācību.
Intranazāla ievadīšana
- Pirms lietošanas pārbaudiet uz primārā iepakojuma norādīto derīguma termiņu. Nelietot ja derīguma termiņš ir beidzies.
- Paņemiet vienu polimēra dozatoru, kas satur NASISOFT šķīdumu, no 10 polimēra dozatoru sloksnes.
- Atveriet polimēra dozatoru, pagriežot tā aizdari ar skrūvējošu kustību.
- Iespiediet šķīdumu no polimēra dozatora deguna atverēs.
Glabāšanas laiks:
2 gadi neskartā iepakojumā.
Kā uzglabāt:
Uzglabājiet atbilstoši noslēgtā iepakojumā temperatūrā no 5 °C līdz 30 °C (ieskaitot), nepakļaujot tiešai gaismas un siltuma avotu iedarbībai. Produkta derīguma termiņš ir spēkā, ja produkts tiek pareizi uzglabāts neskartā iepakojumā.
Iznīcināšana
Produkts jāiznīcina saskaņā ar spēkā esošajiem likumiem par sadzīves atkritumiem.